Any content you receive is for information purposes only. Always conduct your own research. *Sponsored
Jeff Ackerman Just Added AMASS Brands Group (Nasdaq: AMSS) To His Watchlist For This Morning
—Wednesday, June 10, 2026
Don’t Miss Our Next Update—Get Real-Time Alerts Sent Directly To Your Phone. Up To 10X Faster Than Email. Get (AMSS) On Your Radar While It’s Still Early…
June 10, 2026 Pull Up (Nasdaq: AMSS) While It’s Still Early—Last Time it Made an Approx. 110% Move Dear Reader, Did you take a peek at yesterday’s profile? Before the bell even rang, it tapped $.11 — marking an approximate 120% move off of the previous day’s $.05 range. That’s exactly why the Stock News Trends team spends so much time looking for little-known companies arriving on the public markets with real businesses already underneath them. And now, we’re turning our attention to what we’re watching next. AMASS Brands Group (Nasdaq: AMSS) just landed back on our screen this morning—Wednesday, June 10, 2026. But keep in mind, AMSS has less than 10M shares listed as available to the public. When companies have small floats like this, the potential exists for big moves if demand begins to shift. In fact, the last time we highlighted AMSS, it was also a Tuesday night, on May 26, when it closed around $3.58. The very next day, AMSS reached $7.57, capping off an approximate 110% move in less than 24 hours. 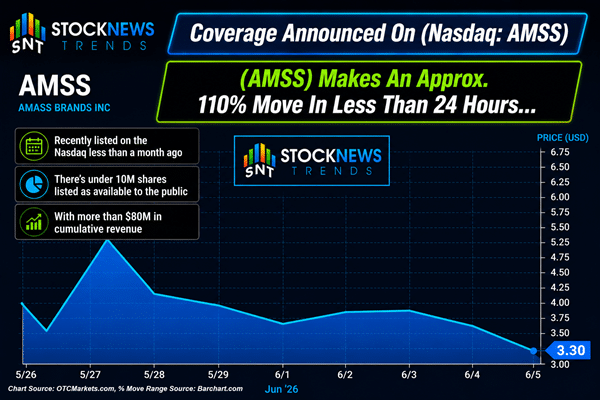
And with AMSS back in focus heading into this morning, the bigger question is what sits underneath the move — and this company has a lot more going on than most newly listed names. Nine premium brands spanning zero-proof wines, wellness-forward functional products, and premium “alcohol 2.0” spirits — all bundled into a single platform that completed its Nasdaq direct listing on May 20, 2026. More than $80M in cumulative revenue, over 5.7M bottles sold, and a distribution footprint already at 40,000+ points of sale — before the street had more than a few days to even process this name. That was compelling enough on its own. But since we first covered AMSS on May 27, 2026, this company has not stopped moving. Three press releases in ten days. Three concrete developments. Each one adding a new layer to a platform that was already built on more operational traction than most newly public companies at this size. Here’s what has happened. What's New at AMASS Brands Group (Nasdaq: AMSS)
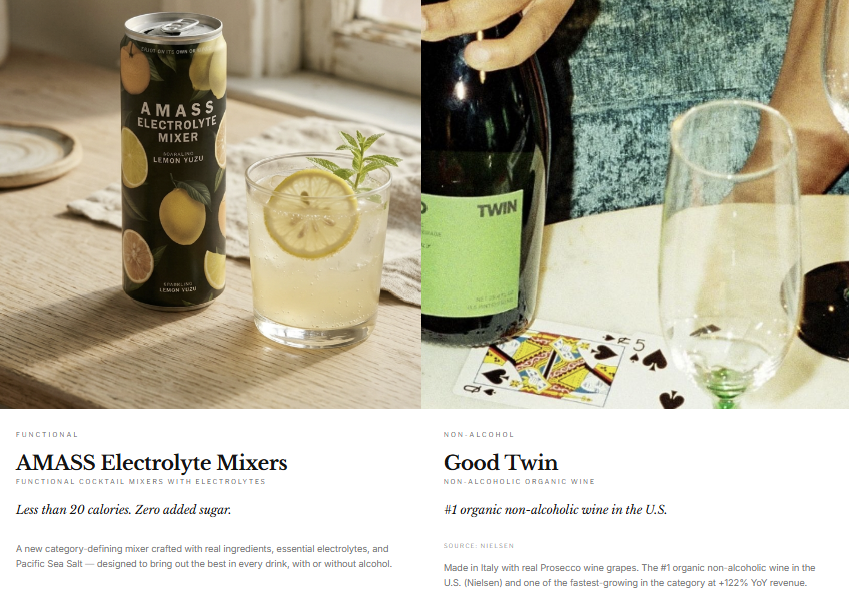
Good Twin Becomes the #1 Organic Non-Alcoholic Wine Brand in the U.S.
On May 27, 2026 — the same morning we first put AMSS on your radar — the company announced that Good Twin Non-Alcoholic Wine had become the #1 ranked organic non-alcoholic wine brand in the United States based on dollar share, according to Nielsen. Good Twin now holds a 35.43% dollar share of the organic non-alcoholic wine category — representing more than one in every three dollars spent in the segment. The brand gained 12.93 share points versus the prior year while delivering +122.2% dollar sales growth and +115.0% growth year-over-year, significantly outperforming the broader organic non-alcoholic wine category, which grew +41.1% during the same period. CEO Mark Thomas Lynn said the company believes it is "still in the early stages of the brand's national expansion.”
AMASS Launches Electrolyte Mixers, Entering the Functional Wellness Category
On May 28, 2026, AMSS announced the launch of AMASS Electrolyte Mixers — the company's first AMASS-branded product in the functional wellness beverage space. Available in ready-to-drink cans, the mixers are formulated with clean-label ingredients, zero added sugar or artificial sweeteners, 20 calories or less per serving, real Pacific sea salt, and essential electrolytes. The product is designed for dual-use — consumed as a standalone wellness beverage or paired as a mixer with alcoholic or non-alcoholic spirits, bridging the functional and social occasion in a single product. The launch arrives at a significant moment for the category. The electrolyte drinks market was estimated at approximately $40B in 2025 and is projected to exceed $80B by 2034, according to Fortune Business Insights. AMASS Electrolyte Mixers are non-GMO, gluten-free, and vegan, and are now available through the company's direct-to-consumer channels and select retail partners, with broader distribution expansion planned throughout 2026. Great Lakes Wine & Spirits Becomes First U.S. Distribution Partner for AMASS Electrolyte Mixers
On June 5, 2026, AMSS announced that Great Lakes Wine & Spirits — Michigan's leading family-owned wholesale alcohol distributor with nearly 80 years of operating history — has become the first U.S. distribution partner for AMASS Electrolyte Mixers. Great Lakes distributes more than 6,000 wine labels, 3,000 spirits, and hundreds of beer and alternative beverage products to retail, on-premise, and hospitality accounts across every county in Michigan. Great Lakes recently launched a dedicated division focused on alternative and better-for-you beverage categories — providing AMASS Electrolyte Mixers with dedicated sales support and focused retail placement efforts from day one. Great Lakes will also carry Summer Water alongside the new electrolyte product, further deepening AMSS's presence in the Michigan market. This partnership establishes the brand's first commercial distribution footprint and marks the beginning of what AMSS is calling a multi-market functional beverage rollout. Inside AMASS Brands Group (Nasdaq: AMSS)

AMSS is a Santa Maria, California-based, multi-category beverage platform founded in 2016 by Mark Thomas Lynn and Morgan McLachlan. The company describes itself as "a next-generation beverage platform built around the brands with the goal of defining how modern consumers drink — and increasingly, how they don't." AMSS operates across three distinct pillars: Non-Alcohol, Functional, and Alcohol 2.0 — a structure the company calls its three-pillar platform, designed to address the full spectrum of the modern social occasion. The portfolio currently spans nine core brands, with standout names including Good Twin Non-Al-coh-olic Wine — now the #1 organic non-alcoholic wine brand in the U.S. with a 35.43% dollar share of its category — and Summer Water Rosé, the zero-sugar, #1 selling premium domestic rosé in the U.S. according to Nielsen, earning 90+ ratings from Wine Enthusiast every year. The broader portfolio also includes Calirosa Tequila, developed in partnership with Adam Levine of Maroon 5; botanical gin and vodka under the AMASS spirits banner; and a range of functional, non-alcoholic spirits designed for consumers who want the social ritual without the alcohol. AMSS distributes through wholesale, on-premise, and direct-to-consumer channels, giving the platform serious omnichannel reach. The company acquired assets from Winc — a direct-to-consumer wine platform that had previously raised $22M in its own IPO — in 2023, adding Summer Water, Pizzolato, and Biokult organic wine brands to its portfolio in a single strategic move. A $314B Functional Beverage Forecast — And AMSS
is Executing Into It…
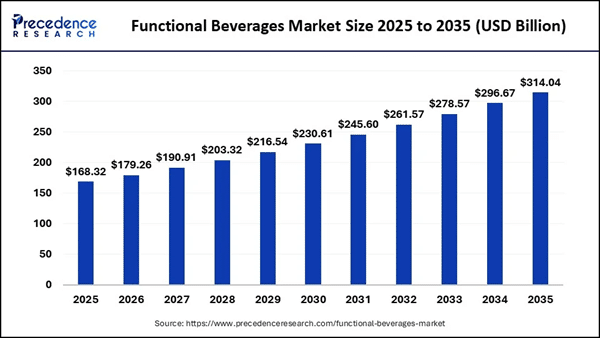
AMSS isn't just building a portfolio of beverages — it's planting a flag at the intersection of two of the fastest-growing segments in the entire consumer staples sector. The global functional beverages market is forecast to expand from roughly $179B in 2026 to over $314B by 2035. Meanwhile, the U.S. non-alcoholic beverages market is projected to surpass $246B by 2032, up from approximately $169.6B in 2024. And the electrolyte drinks market alone is on track to grow from roughly $40B in 2025 to over $80B by 2034. These are not niche tailwinds. They represent a fundamental generational reshaping of how Americans consume beverages — and AMSS now has active, commercial product lines running inside all three of those trajectories simultaneously. The "sober curious" and moderation movements have moved well beyond trend status; they now represent a structural shift in consumer behavior, particularly among younger demographics. AMSS has positioned its entire architecture to benefit from this shift without abandoning the premium economics of traditional alcohol — a balance that gives it staying power across market cycles. Multi-Brand Platform = Built-In Scale and M&A Potential
What makes AMSS structurally differentiated is that it is not built around a single brand. The company's core strategy centers on a "cohesive brand architecture designed for both organic scale and relevance to modern beverage acquirers." Translation: the platform is explicitly built to be attractive to a larger strategic buyer, while also being capable of scaling each brand independently. The company has demonstrated an appetite for smart acquisitions — the Winc asset purchase being a case in point — and management has signaled continued focus on both organic growth and strategic M&A as public company tools. With distribution already in place at 40,000+ points of sale, any incremental brand addition lands on an established commercial backbone. A Newly Public Beverage Platform Built Around Scale
and Discipline
AMSS chose a direct listing structure rather than a traditional IPO, meaning the company did not issue new shares or dilute existing holders in connection with going public. CEO Mark Thomas Lynn has been direct about the company's ambitions since day one, saying: "We've built a differentiated, multi-brand platform positioned at the intersection of several of the fastest-growing segments in the beverage industry," Lynn said in the company's Nasdaq listing announcement. As a public company, he added, the focus is on scaling the portfolio, expanding distribution, and executing on the strategy to build a leading next-generation beverage company. 7 Reasons Why AMSS is Back at the Top of Our Watchlist
This Morning—Wednesday, June 10, 2026…
1. Low Float: With less than 10M shares listed as available to the public, AMSS’s small float could witness the potential for big moves if demand begins to shift. 2. Recent Momentum: AMSS recently went from around $3.58 to $7.57 in less than 24 hours, representing an approximate 110% move. 3. Fresh Listing: AMSS completed its Nasdaq direct listing on May 20, 2026, making it one of the newer names on the exchange. 4. Established Revenue: AMSS reported more than $80M in cumulative revenue before many market participants had fully become familiar with the company. 5. Nine Brands: AMSS operates a portfolio of nine beverage brands spanning non-alcoholic, functional wellness, and premium spirits categories. 6. Category Leader: AMSS owns Good Twin, which was ranked the #1 organic non-alcoholic wine brand in the United States by dollar share according to Nielsen. 7. Active News Flow: AMSS announced three separate developments within ten days, including a new product launch, a category leadership milestone, and its first U.S. distribution partnership for AMASS Electrolyte Mixers. Get (AMSS) On Your Radar While It’s Still Early…

When we covered AMASS Brands Group (Nasdaq: AMSS), the case rested on a fresh Nasdaq listing, a tight public float, strong underlying fundamentals, and a multi-brand platform sitting inside powerful category tailwinds. That case has only gotten stronger. Since we first covered AMSS, the company has announced that Good Twin has become the undisputed leader in U.S. organic non-alcoholic wine — holding a 35.43% dollar share of the category and delivering 122.2% year-over-year dollar sales growth according to Nielsen data through April 18, 2026. It has launched a brand new functional wellness product — AMASS Electrolyte Mixers — into a category projected to reach $80B by 2034. And it has secured its first commercial distribution partner for that product in Great Lakes Wine & Spirits, one of the Midwest's most established distribution networks with nearly 80 years of regional presence. This is a company that hit the ground running the moment it went public. Take a look at AMSS this morning while it’s still early. And keep an eye out for my next update, it could be coming very shortly.
Sincerely, Jeff Ackerman Managing Editor Stock News Trends |