Any content you receive is for information purposes only. Always conduct your own research. *Sponsored
For This Morning, bioAffinity Technologies, Inc. (Nasdaq: BIAF) Is Officially on Market Crux's Radar—Tuesday, March 10, 2026
Don't Miss The Next Breakout—Get Real-Time Alerts Sent Directly
To Your Phone. Up To 10X Faster Than Email.
We're Kicking Off Full Coverage On (BIAF) Today
Get (BIAF) On Your Radar While It's Still Early…
March 10, 2026
New Development | (BIAF) Pops Approx. 39% On Breaking Headline
Dear Reader, Early developments surrounding bioAffinity Technologies, Inc. (Nasdaq: BIAF) are beginning to stir interest this morning, and Market Crux is already on it. Shares of (BIAF) already tapped $1.39, marking an approximate 39% move from yesterday's $1 range as fresh headlines began circulating. At the same time, TradingView's technical analysis dashboard is lighting up with multiple bullish signals, putting the company squarely back on the radar for momentum-focused traders. The catalyst behind the move is a newly announced large-scale longitudinal clinical study for CyPath® Lung, the company's noninvasive diagnostic designed to help detect early-stage lung cancer. The planned 2,000-patient study will evaluate the clinical performance of the sputum-based flow cytometry test in high-risk individuals with existing lung nodules, with participation expected from up to 20 clinical sites, including several Department of Veterans Affairs medical centers and major military hospitals. Support for the study is being provided by the Murtha Cancer Center Research Program at the Uniformed Services University of the Health Sciences, adding institutional backing as the company seeks to generate additional real-world clinical data around CyPath® Lung. Management says the study could provide further evidence supporting the inclusion of this noninvasive test in the standard of care for lung cancer screening and diagnosis, particularly as physicians look for ways to evaluate indeterminate lung nodules without immediately resorting to invasive procedures. With the announcement now circulating and (BIAF) already showing its potential for big swings, this story is at the top of our screen. If you missed my earlier coverage email, keep reading to quickly get up to speed on why we're so excited to be highlighting (BIAF) today. ===== Market Crux keeps a close watch on little-known companies tied to big shifts that can quickly move into focus. One of the clearest examples developing right now is in lung cancer screening, where noninvasive diagnostic technology is starting to reshape how physicians handle one of the toughest decisions in clinical care. For years, patients with suspicious lung nodules have often faced repeat imaging, lingering uncertainty, or invasive procedures that ultimately reveal no cancer was present. That dynamic is beginning to change as more advanced tools move out of research environments and into real-world clinical use, giving physicians a more precise way to evaluate risk earlier in the process. As this transition continues to build, companies bringing practical diagnostic solutions to the front lines of healthcare are beginning to draw more attention. bioAffinity Technologies, Inc. (Nasdaq: BIAF) is one of the companies operating within this shift, using flow cytometry and proprietary analysis to support the detection of early-stage malignancy. That is exactly why (BIAF) just moved onto my radar and is topping my watchlist this morning—Tuesday, March 10, 2026.
But keep in mind, (BIAF) has less than 5M shares listed as available to the public, according to MarketWatch. When companies have small public floats like this, the potential exists for big moves if demand begins to shift. This is already starting to show on the chart. (BIAF) recently made an approximate 90% move in under two weeks, from around $.73 on February 6 to $1.39 on February 19, according to data available from Barchart. 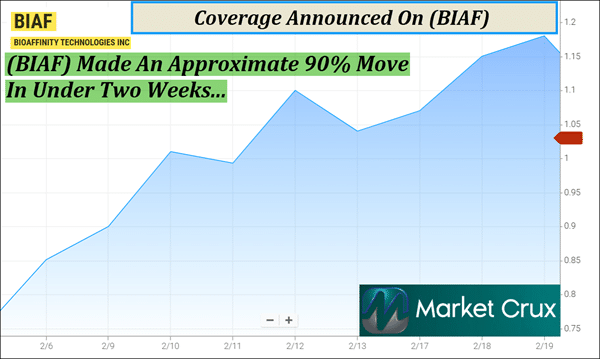
While traditional methods often lead to invasive procedures for benign findings, the company is advancing a science-based alternative designed to improve decision-making in lung cancer diagnostics. And the deeper you look into what (BIAF) is building, the easier it becomes to see why this company is starting to draw attention. See the company's latest corporate presentation here. Here's a closer look at the technology, clinical utility, and real-world factors that are putting (BIAF) on our radar. Transforming Early Detection
bioAffinity Technologies, Inc. (Nasdaq: BIAF) is a biotechnology company focused on the noninvasive diagnosis of early-stage cancer and other diseases of the lung. The company's flagship product, CyPath® Lung, addresses a critical gap in the current healthcare system: the high rate of false positives in lung cancer screening. When a low-dose computed tomography (LDCT) scan identifies a suspicious pulmonary nodule, physicians are often forced to choose between "watchful waiting" or aggressive, invasive biopsies. CyPath® Lung changes this dynamic by analyzing a patient's sputum (phlegm) to identify cell populations indicative of cancer. The test is currently marketed as a Laboratory Developed Test (LDT) through the company's subsidiary, Precision Pathology Laboratory Services. This facility recently maintained its prestigious College of American Pathologists (CAP) accreditation, which is considered the gold standard for laboratory excellence and accuracy. By providing a 92% sensitivity and 87% specificity for detecting cancer in nodules smaller than 2 centimeters, (BIAF) is delivering a high-accuracy tool that helps physicians rule out cancer without surgery. The $10.4B Market
The U.S. lung cancer diagnostics market is projected to more than double within the next decade to reach $10.4B by 2034. This projected growth is being driven in part by a rising emphasis on early detection, as lung cancer remains the leading cause of cancer-related death worldwide. Current screening guidelines recommend annual LDCT scans for millions of high-risk individuals, yet uncertainty around scan results continues to create a significant diagnostic burden for both patients and physicians. bioAffinity Technologies, Inc. (Nasdaq: BIAF) is developing CyPath® Lung to address this challenge by providing additional clinical data after a suspicious nodule is identified. 
In recent case material, the test supported a conservative surveillance strategy for an elderly patient with a suspicious nodule. A later follow-up scan showed that the nodule had resolved, supporting the decision to avoid an invasive and costly biopsy. The ability to help rule out cancer with a high degree of confidence could become an important point of differentiation in a field where false positives often lead to unnecessary medical intervention. Platform Versatility: From Oncology to Asthma
Beyond its core lung cancer application, (BIAF) is proving that its flow cytometry platform has broad-spectrum potential. On March 3, 2026, the company presented positive research at the American Academy of Allergy, Asthma & Immunology (AAAAI) Annual Meeting. The data demonstrated the technology's ability to identify specific antibody receptors in sputum for leading asthma therapies, including dupilumab and benralizumab. This breakthrough suggests that the (BIAF) platform can be used to identify optimal therapies for the approximately 650M children and adults globally suffering from asthma and COPD. By matching patients with the most effective biologic therapies and monitoring inflammatory changes over time, (BIAF) is opening a new vertical in personalized medicine. This diversification reduces reliance on a single diagnostic area and positions the company as a leader in respiratory health technology. Recent Operational Achievements
Operational achievements point toward increased accessibility and commercial scale. The company recently announced a validation study with Brooke Army Medical Center (BAMC) to expand sample collection options. By validating deep suction collection alongside spontaneous cough samples, (BIAF) could significantly accelerate the adoption of CyPath® Lung in clinical settings where patients may struggle to produce a sample naturally. Results from this study are expected by mid-year 2026. Furthermore, the company's financial results for the third quarter of 2025 showcased a strategic pivot. While total revenues were $1.4M, the company reported a 122% year-over-year increase in CyPath® Lung testing revenue. This growth was driven by higher case volumes and increased adoption within the Veterans Administration (VA) health system. The company also significantly improved its balance sheet, ending the period with $7.7M in ca-sh, up from $1.1M at the end of 2024. Management and Advisory Strength
The leadership team at (BIAF) is led by Maria Zannes, a veteran executive with over 30 years of experience. She has built a team of award-winning scientists, including Chief Science Officer Dr. William Bauta, a medicinal chemist with 25 years of experience in dr-ug discovery and FDA approvals. To further guide its commercial strategy, the company recently appointed nationally recognized authorities to its Medical and Scientific Advisory Board, including specialists from MD Anderson Cancer Center and NYU Grossman School of Medicine. These experts provide independent guidance on clinical priorities and help drive the integration of CyPath® Lung into the standard of care. 7 Factors Putting (BIAF) At The Top Of This Morning's Watchlist
—Tuesday, March 10, 2026…
1. Recent Momentum: Chart data from Barchart shows that (BIAF) made an approximate 90% move in under two weeks, from about $0.73 on February 6 to $1.39 on February 19. 2. Small Float: With fewer than 5M shares listed as available to the public, (BIAF)'s small float could have the potential for big moves if demand begins to shift. 3. Growing Market: Industry projections indicate the U.S. lung cancer diagnostics market is expected to more than double to about $10.4B by 2034, placing (BIAF) within a rapidly expanding segment of healthcare diagnostics.
4. Noninvasive Detection: Through its CyPath® Lung test, (BIAF) is developing a sputum-based method designed to help identify early-stage lung malignancy without the need for invasive diagnostic procedures. 5. Operational Progress: Recent financial disclosures show that (BIAF) reported a 122% year-over-year increase in CyPath® Lung testing revenue while strengthening its balance sheet with $7.7M in ca-sh at the end of Q3 2025. 6. Platform Expansion: Research presented at the AAAAI Annual Meeting suggests the technology platform developed by (BIAF) may help identify antibody receptor responses related to leading asthma therapies. 7. High Accuracy: Clinical data cited for CyPath® Lung shows that (BIAF) achieved 92% sensitivity and 87% specificity when detecting cancer in nodules smaller than 2 centimeters. Get (BIAF) On Your Radar While It's Still Early…

(BIAF) just hit our radar for several reasons. The company has fewer than 5M shares available to the public, and small floats like this can sometimes have the potential for big moves if demand begins to shift. Recent chart activity already shows signs of momentum, with (BIAF) recently making an approximate 90% move in under two weeks. At the same time, the company is working within a lung cancer diagnostics market that industry projections suggest could more than double to about $10.4B by 2034. Beyond the market backdrop, the underlying technology is what makes this story worth understanding. CyPath® Lung is designed as a noninvasive sputum-based test that helps physicians evaluate suspicious lung nodules without immediately resorting to invasive procedures. Clinical data cited for the test reports 92% sensitivity and 87% specificity when identifying cancer in nodules smaller than 2 centimeters. On top of that, research presented earlier this month suggests the same platform may help identify antibody receptor responses connected to leading asthma therapies. Operational progress is also beginning to appear in the numbers. CyPath® Lung testing revenue increased 122% year-over-year, and the company reported $7.7M in ca-sh at the end of Q3 2025. (BIAF) is the only name we're watching this morning—Tuesday, March 10, 2026. Get (BIAF) on your radar while it's still early, and be on the lookout for my next update, it could be coming very shortly. Sincerely, Gary Silver
Managing Editor,
Market Crux
| 



0 التعليقات:
إرسال تعليق